ABOUT PROJECT REAP:
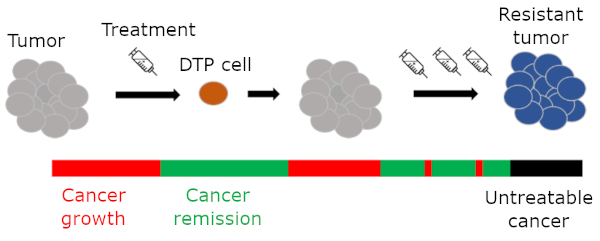
Breast cancer remains the second most common cause of cancer-related death in women. Available therapies, which are initially effective, lose potency as cancer cells regrow and new tumors appear. These new tumors can originate from a type of drug tolerant persister (DTP) cells, which evolve in response to therapy. As these cells are not affected by the administered drugs, they persist after most cancer cells are eliminated and act as sources for new tumors when treatment is stopped. What is particularly troublesome about DTP cells is that there are no effective methods of detecting them. Project REAP intends to change that.
The presence of DTP cells ensures the generation of new tumors which evolve to resist treatment over multiple courses of therapy.
Ultrasound, magnetic resonance imaging, and mammography are effective detection techniques typically used to diagnose breast cancer tumors. However, they fail at detecting these stay-behind cells. This presents a unique challenge to current cancer detection technologies, and a need for the development of new ones specifically targeting DTP cells.
Each of the imaging systems will have a different target: an optical coherence photoacoustic microscopy (OC-PAM) system to make images of cancer samples at cellular and sub-cellular levels, an optical coherence photoacoustic tomography (OC-PAT) system to make images of very small tumor clusters and their environments, and a second PAT system to make images of the whole body to track tumor growth and whether it has moved to new organs.
THE ROLE OF LIONIX INTERNATIONAL:
While we have experience working on integrated optical coherence tomography systems, it is our tunable lasers that we are contributing to this project. Our role is to develop high-speed tunable laser sources to be used in the photoacoustic microscopy and tomography systems.
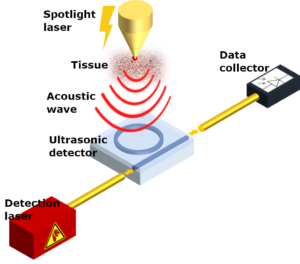
Photoacoustic imaging uses lasers to heat up a section of organic tissue. This increase in temperature causes the emission of acoustic waves which are picked up by ultrasonic detectors. Lasers are also used in these ultrasonic detectors to identify the acoustic wave, making the quality of these lasers crucial to the entire diagnostic procedure of the device.
The linewidth of the light that a laser generates is paramount to its quality. A laser with a narrow linewidth ensures that the maximum amount of power is dedicated towards producing the intended range of wavelengths. In this project, a tunable laser is required, where the output wavelength of the laser can be adjusted.
Our tunable lasers provide exceptionally narrow linewidths (<10 kHz) with impressive power outputs (>20 mW). This performance is combined with wide-range tunability (40 nm) and high-speed operation (<1 msec), all in a tiny package. The laser is made possible using the hybrid integration of an InP gain chip and a TriPleX® modulation chip, making the external cavity laser the best of both passive and active photonics worlds.
A general schematic of the instruments used in photoacoustic spectroscopy. LioniX will be involved in the development of the detection lasers shown in red.
The laser used in this application will be centered around 780 nm, a customized version of our 1550 nm laser. The laser will feature 40 nm tunability around the 780 nm central frequency at <1 millisecond tuning speed. The narrow linewidth provided by the laser is crucial for the proper imaging of the target tissue. As no affordable compact tunable lasers at 780 nm exist today, our contribution is a true breakthrough. The laser will act as a light source for both microscopy and tomography systems, significantly reducing the cost and size of the total device. This development is carried out in close collaboration with our project partners and under the supervision of the European Commission. We look forward to sharing the fruits of this labor with the public at the end of the project duration.
Further information:
For more information about REAP, please visit the project website here.
For a summary of the project and our 780 nm laser, download our briefing here.